  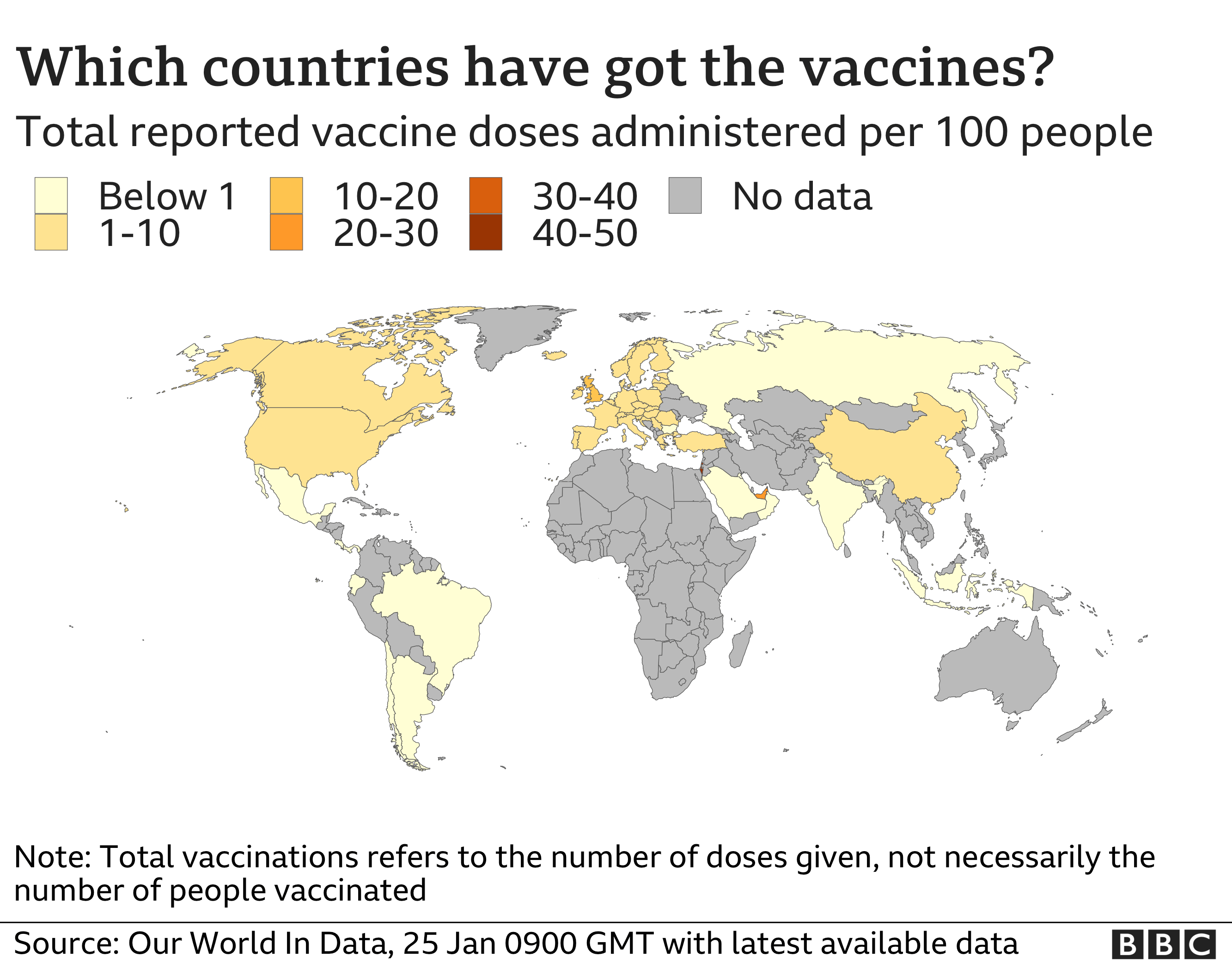
This is the third COVID-19 vaccine that EMA has recommended for authorisation. "Member states could also extend this to those vaccinated with a vaccine having completed the WHO emergency use listing process," an official statement added. EMA has recommended granting a conditional marketing authorisation for COVID-19 Vaccine AstraZeneca to prevent coronavirus disease 2019 (COVID-19) in people from 18 years of age. The proposals were first published by the European Union earlier this month after bloc member Greece announced that it would be welcoming fully vaccinated or COVID-tested international travelers in the coming weeks. Emergency brake optionĪn EU official told CNN that the bloc will take reciprocity into account when drawing up its list, but recommendations suggest that countries with less than 75 cases per 100,000 people will be included.Ī formal adoption of the recommendations - which are not legally binding for member states - is expected to happen on Thursday. Vaccine deliveries to EU countries have increased steadily and vaccination is gathering pace. The EMA hasn't approved any vaccines from Russia or China as of yet but is looking at data for Russia's Sputnik V jab. So in short, mixed dose vaccines are fine to enter France with, but bear in mind the other criteria to fulfil before you can be accepted as fully vaccinated. Safe COVID-19 vaccines for Europeans The European Commission has secured up to 4.2 billion doses of COVID-19 vaccines so far and negotiations are underway for additional doses. However, the plans allow for an "emergency brake" in case infection rates rise again.Ĭoronavirus vaccines authorized by the European Medicines Agency, the bloc's drug regulator, include Pfizer, Moderna, AstraZeneca and Johnson & Johnson. The decision comes on the back of the European Medicines Agency (EMA) authorization, which was announced yesterday. Officials are said to be optimistic that the new rules will come into effect in June, providing a much-needed boost to the tourism industry as summertime begins. Covid-19 vaccines EU drug agency recommends approval of Covid vaccine boosters for Omicron August 7 2022. The World Health Organization (WHO) today listed the COVID-19 vaccine, developed by Janssen (Johnson & Johnson), for emergency use in all countries and for COVAX roll-out. It's thought that individual member states will have the final decision on what measures to impose, which means some could still opt to keep quarantine measures in place, while others may ask for negative PCR tests and/or proof of vaccination. The highly anticipated move was confirmed on Wednesday as the EU published its recommendations for lifting travel restrictions into the bloc.Īn approved list of "safe" destinations is due to be signed off this week, although there's still no confirmation as to when these changes will be implemented. The European Union has taken a step toward relaxing travel for visitors from outside the bloc, making it easier for fully vaccinated visitors to get in.īRUSSELS - Almost a year after closing its borders, the European Union has agreed to allow entry to vaccinated travelers from countries with low infection rates, raising the possibility of summer getaways to the continent. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
